Epigenomics and cancer heterogeneity in Ewing sarcoma
by Nathan Sheffield
Center for Public Health GenomicsUniversity of Virginia
Today's outline

Epigenomics and cancer heterogeneity in Ewing sarcoma
Ewing sarcoma
 |
Rare, aggressive pediatric bone cancer |
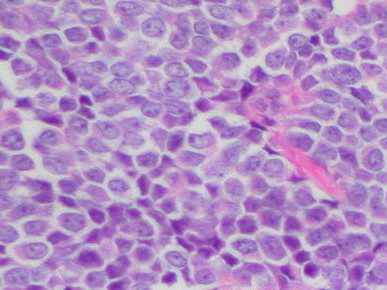 |
Small cell tumor neuroblastoma, Wilms' tumor, lymphoma desmoplastic small-round-cell tumor |
 |
Driven by a single, well-characterized oncogenic fusion transcription factor (EWS-FLI1) |





Epigenomics and cancer heterogeneity in Ewing sarcoma
Ewing sarcoma clinical heterogeneity
- Ewing (Ewing 1921) vs. Peripheral primitive neuro-ectodermal tumor (pPNETs; Stout 1918)
- osseous vs. extra-osseous (Tefft et al. 1969)
- site of origin: Askin tumor of the chest wall (Askin et al. 1979)
"ES and pPNET are considered as two tumor phenotypes along a gradient of limited neuroglial maturation that arise from the same stem cell” (Kovar 2005)
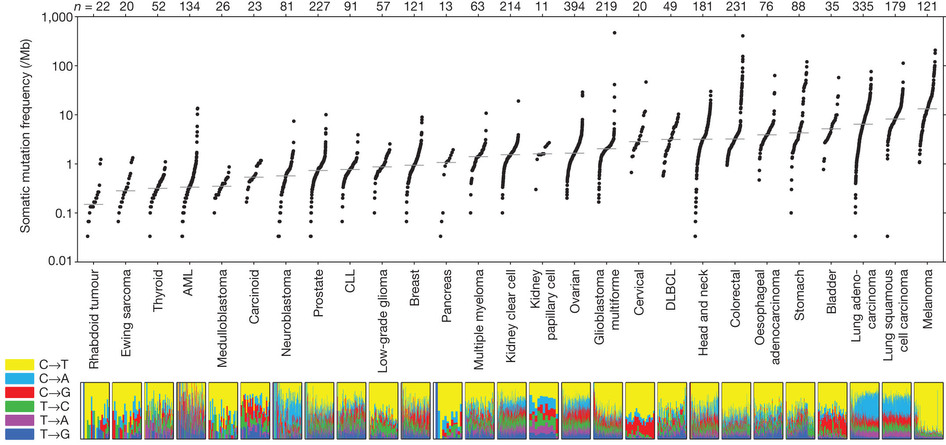 Lawrence et al. 2013
Lawrence et al. 2013

RRBS dataset
- 140 primary Ewing sarcoma tumors
- 32 primary mesenchymal stem cell samples (3 sources: bone marrow, umbilical cord, placenta)
- 16 Ewing sarcoma cell lines (6 derived from these tumors)
RRBS reference panel
- Cancer comparison: 105 samples from 7 other cancer types
- Cell-type diversity: 266 samples from 50+ tissues
Why RRBS?
Nice cost/benefit ratioSingle-nucleotide resolution

Acknowledgments
St. Anna's Child Cancer Research Institute, Vienna
Heinrich KovarEleni Tomazou
Peter Ambros
Inge Ambros
Diana Walder
Paracelsus Medical University, Salzburg
Dirk StrunkKatharina Schallmoser
Medical University of Graz
Beate RinnerBernadette Liegl-Atzwanger
Berthold Huppertz
Andreas Leithner
CeMM Research Center for Molecular Medicine, Vienna
Christoph BockJohanna Klughammer
Andreas Schönegger
Michael Schuster
Paul Datlinger
Johanna Hadler
Münster University Hospital
Uta DirksenInstitute of Biomedicine of Seville, Spain
Ana T. AmaralEnrique de Álava
Institut Curie, Paris
Olivier DelattreFranck Tirode
Sandrine Grossetete
Funding
Human Frontier Science ProgramKapsch NGS Grant
FWF Lise Meitner Fellowship





















Proportion Intermediate Methylation






- DNA methylation quantifies cancer heterogeneity
- DNA methylation can be used to infer regulatory activity
- EWS-FLI suppresses mesenchymal regulatory activity
- Mesenchymal regulatory elements are more suppressed in STAG2-mutants
- More heterogeneity (higher PIM) signals cancer progression
Lightning ideas
Bioinformatics pipeline framework

Chromatin dynamics in mitosis

Single-cell omics data analysis

Locus Overlap Analysis (LOLA)

HiChIP - 3d interactions
Questions?
nsheffield@virginia.edugithub.com/nsheff
databio.org